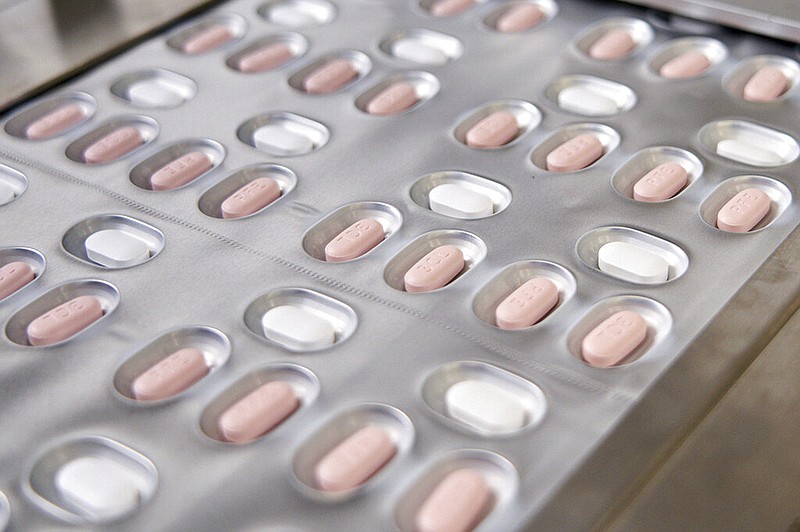
WASHINGTON -- Pharmacists may prescribe the leading covid-19 pill directly to patients under a new U.S. policy announced Wednesday that's intended to expand use of Pfizer's drug Paxlovid.
The Food and Drug Administration said pharmacists may begin screening patients to see if they are eligible for Paxlovid and then prescribe the medication, which has been shown to curb the worst effects of the virus. Previously only physicians could prescribe the antiviral drug.
The announcement comes as covid-19 cases, hospitalizations and deaths are rising again, though they remain near their lowest levels since the pandemic began in 2020.
Biden administration officials have expressed frustration that several hundred Americans continue to die of covid-19 each day despite the availability of vaccines and treatments.
Administration officials have been working for months to increase access to Paxlovid, opening thousands of sites where patients who test positive can fill a prescription for Paxlovid. The FDA change will make thousands more pharmacies eligible to quickly prescribe and dispense the pill, which must be used early to be effective.
"Since Paxlovid must be taken within five days after symptoms begin, authorizing state-licensed pharmacists to prescribe Paxlovid could expand access to timely treatment," FDA drug center director Patrizia Cavazzoni said.
Still, use could be limited by paperwork requirements. Patients are expected to bring their recent health records -- including blood tests -- and a list of their current medications so pharmacists can check for health conditions and medications that can interact with Paxlovid. As an alternative, pharmacists can consult with the patient's doctor.
Paxlovid is intended for people with covid-19 who are more likely to become seriously ill. That includes older people and those with health issues such as heart disease, obesity, cancer or diabetes that make them more vulnerable. It isn't recommended for patients with severe kidney or liver problems. A course of treatment is three pills twice a day for five days.
The FDA authorized Paxlovid in December for people 12 and older based on results showing that it reduced hospitalizations and deaths by nearly 90% among unvaccinated patients most likely to get severe disease. The drug has shown less impressive results in patients who already have vaccine protection, and some physicians have reported cases of covid-19 symptoms returning after treatment with the drug.
THOUSANDS OF OUTLETS
Expanding the test-to-treat program to include pharmacists could add thousands of additional options for patients. The two biggest U.S. drugstore chains -- CVS Health and Walgreens -- have about 19,000 locations combined.
CVS already provides covid-19 care at 1,100 clinics in drugstores.
There also are nearly 19,400 independent pharmacies not tied to a big chain, according to the National Community Pharmacists Association.
Pharmacist Michele Belcher said she hoped to be able to test customers for covid-19 and offer the pill because there is a shortage of primary care doctors in her community, the small, southwestern Oregon city of Grants Pass.
Belcher said she worries that some people may have a hard time getting a doctor's appointment for a prescription in the narrow window to start the pill.
Belcher, owner of the independent Grants Pass Pharmacy, said she used to test and treat for covid-19 using injectable drugs that are no longer as effective.
Her pharmacy routinely checks for potentially harmful interactions with other drugs a patient may be taking, she said.
"Pharmacists are the drug experts," she said. "That's something we do every day, all day, make sure there are no interactions with any medications."
NEW DRUG OFFERS HOPE
Meanwhile, an experimental drug initially developed to fight cancer cut the risk of death for people hospitalized with covid by half, according to a study published Wednesday.
The drug, sabizabulin, seemed to be more effective than others that have been authorized for severely ill covid patients.
Veru, the company in Miami that developed the drug, has applied to the Food and Drug Administration for emergency authorization. That would add a new weapon to the modest arsenal available to hospitalized patients, experts said.
"This looks super-impressive," said Dr. Ilan Schwartz, an infectious disease expert at the University of Alberta who was not involved in the study. "We have a small number of treatments for patients with severe disease that improve mortality, but another treatment that can further reduce deaths would be very welcome."
Schwartz cautioned, however, that the trial was relatively small, with just 134 patients taking the drug. "Overall, I think this is very exciting, although I would welcome larger and independent confirmatory studies," he said.
Sabizabulin blocks cells from building microtubules, critical molecular cables that shuttle material from one part of the cell's interior to another.
The drug was developed by researchers at the University of Tennessee to fight cancer, as tumor cells depend on the microtubules for their rapid growth.
Two years ago, researchers at Veru tried sabizabulin on covid. They suspected it might prevent viral replication, which depends on the microtubule network to bring together the pieces of new viruses.
They also hypothesized that the drug would help covid patients fight potentially life-threatening lung inflammation. This immune response starts when cells recognize they are infected and release alarm-signal proteins into their surroundings. The cells have to push the alarm molecules along their microtubules to get the word out.
134 VOLUNTEERS
In early 2020, researchers at the University of Tennessee Health Science Center found that sabizabulin tamped down these alarm signals in mouse cells. A few months later, Veru began testing the drug, which is taken as a pill, in people. In May 2021, it advanced to a late-stage trial.
The company sought volunteers who were already hospitalized for covid. To be eligible for the trial, the patients had to be receiving oxygen or relying on a ventilator. They also had to be at high risk of dying of covid, with risk factors such as hypertension, advanced age or obesity.
The patients were allowed to simultaneously receive other treatments that have been shown to be effective at saving lives of hospitalized covid patients. A steroid called dexamethasone, for example, reduces the risk of death by one-third.
In the latest trial, 134 volunteers received sabizabulin and 70 a placebo. Over the course of 60 days, the death rates of the two groups were significantly different: 45.1% of the placebo group died, compared with just 20.2% of those who received the new drug. That difference translated to a 55.2% reduction in the risk of death.
Dr. David Boulware, an infectious disease expert at the University of Minnesota, cautioned that the large number of deaths in the placebo group could be a sign the study was too small to draw firm conclusions.
"The 45% mortality rate in the control group jumps out at me as rather high," he said.
A number of antiviral drugs have proved effective at keeping covid patients out of the hospital, but only if they are given early in the course of their disease. Paxlovid, for instance, can reduce the risk of hospitalization for unvaccinated people with covid risk factors by about 90%.
These drugs do not work well, however, on hospitalized patients with moderate to severe covid. That's because they only block viruses rather than reining in the runaway immune system's response.
For hospitalized patients, doctors have fewer drugs to choose from. In addition to dexamethasone and baricitinib, another anti-inflammatory drug called tocilizumab has been shown to help.
When Veru initially announced its results in April, it said it halted the trial early because an independent advisory committee found the benefits of sabizabulin were already clear from the data; it would be unethical, they decided, to keep giving some patients a placebo.
While Boulware recognized the ethical demands of the situation, he also predicted that if the trial had gone longer, the benefits of the drug might have emerged as more modest.
Information for this article was contributed by Matthew Perrone and Tom Murphy of The Associated Press, and by Carl Zimmer of The New York Times.
 A boy holding a giant mock syringe that reads “sovereignty” in Spanish takes part in a protest Wednesday in Montevideo, Uruguay, outside the Supreme Court during a hearing where the government and Pfizer gave more detailed information on covid-19 vaccinations for children. Vaccinations of children over age 5 in the country are voluntary. (AP/Matilde Campodonico)
A boy holding a giant mock syringe that reads “sovereignty” in Spanish takes part in a protest Wednesday in Montevideo, Uruguay, outside the Supreme Court during a hearing where the government and Pfizer gave more detailed information on covid-19 vaccinations for children. Vaccinations of children over age 5 in the country are voluntary. (AP/Matilde Campodonico)